|
1/12/2024 0 Comments Number 9 on periodic table 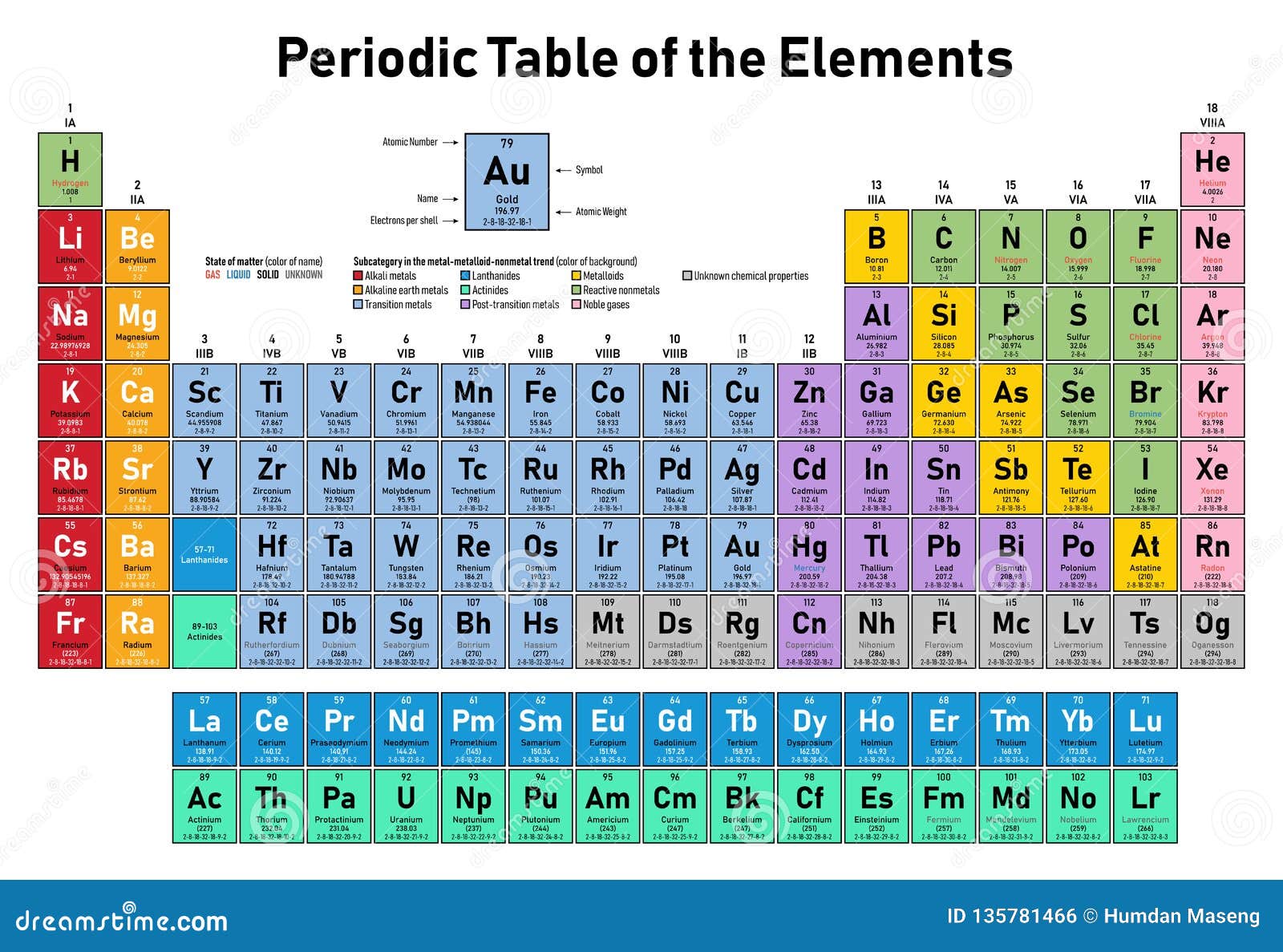
Polymeric perfluorinated ethers are widely used as high performance oils and lubricants. Polytetrafluoroethylene can be modified to form a coating, Gore-Tex, which allows the passage of water vapor, but not liquid water, and is used in many articles of clothing. Teflon (polytetrafluoroethylene) is a chemically inert polymer used to create nonstick frying pans. Substitutes for these ozone-depleting compounds are being developed. Low molecular weight chlorofluorocarbons, known as Freons, are nonflammable, dense, nontoxic compounds particularly useful as refrigerants and blowing agents. The new compounds have increased chemical, thermal, and oxidative stability. When hydrogen is replaced by fluorine in organic compounds, the properties of the compounds are substantially altered. Sodium fluoride, NaF, is often added to drinking water and to toothpaste in order to reduce the incidence of tooth decay. Other important inorganic compounds of fluorine include uranium hexafluoride, UF 6, a volatile substance that was used to separate 238U from the fissionable 235U by gaseous diffusion in the Manhattan Project. These minerals are widely used as electrolytes in the preparation of aluminum, as fluxes in the metallurgy of iron, and as additives in ceramics. In nature, fluorine is primarily found in the minerals fluorospar (CaF 2), cryolite (Na 3AlF 6), and fluoroapatite (CaF 2 ♳Ca 3(PO 4) 2), together comprising 0.065 percent of Earth's crust (making fluorine the thirteenth most abundant element). Fluorine forms compounds with all of the elements except helium, neon, and argon. The low F –F bond energy and the high energy of bonds between fluorine and other elements combine to make reactions of fluorine exothermic.Įlemental fluorine was first prepared by Henri Moissan in France in 1886 via the electrolysis of anhydrous hydrogen fluoride in the presence of potassium fluoride. It is the most powerful oxidizing agent known. 
Fluorine is a poisonous, corrosive, pale yellow gas with an acrid odor. It is the lightest and most reactive element in the halogen family. The inclusion of the isotope 18F (half-life 110 minutes) in bioorganic molecules is an important noninvasive technique used in the study of living tissue by positron emission tomography.įluorine is the most electronegative element. However, the radioactive isotopes 17F, 18F, and 20F are known. There is only one stable, naturally occurring isotope of fluorine: 19F. The fluorine atom has the ground state electron configuration 1s 22s 22p 5. Melting point: −223 ☌ boiling point: −188 ☌ density: 1.696 g/cm 3 most common ions: F −įluorine is a chemical element that in pure form occurs as a dimer of two fluorine atoms, F 2. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
